No products in the cart.
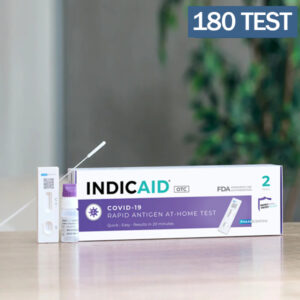
Indicaid OTC Antigen Rapid Test Kit 126 Kits (252 test)
$1,500.00 $850.00
Indicaid OTC Antigen Rapid Test Kit (252 test per case), 126 Kits of 2-count, IN STOCK.
Please send inquires for volume pricing.

SKU: Indicaid-POC-test-25pc
Categories: All Products, Medical Equipments, Wholesale
Tags: covid sars antigen test, covid test kit los angeles wholesale cheap bulk volume, indicaid poc antigen rapid test
Indicaid OTC Antigen Rapid Test Kit 126 Kits (252 test)
OTC refers to Over-The-Counter. This listing is by the case of 126 kits (2 test per kit), equivalent to 252 tests.
We also carry the other OTC 12 Per Kit version. If interested please send us a message at order@healthsupplyworld.com
These items are available stock, for volume pricing please send inquiries.
Expiration dates are good (not expired) so it’s safe to use for general use.
FDA Emergency Use Authorization
The Indicaid OTC Antigen Rapid Test has not been FDA approved, however it has been authorized by the FDA under the Emergency Use Authorization (EUA) for use by authorized laboratories for the defection of proteins from SARS-CoV-2, not for any other pathogens or viruses. Provides accurate reliable results within ten minutes, allowing for symptomatic patients within the first five days of symptoms.
- Results within ten minutes
- Ready to use
- Please follow instructions on package
- No refrigerator space needed.
- Test can be run immediately as needed
For information please go to: https://us.phasescientific.com/indicaid-rapid-antigen-test/#
We also supply other brands of Covid test kits for Healthcare professionals and other Healthcare Brokers.
Be the first to review “Indicaid OTC Antigen Rapid Test Kit 126 Kits (252 test)” Cancel reply
Related products
Sale!
Sale!
Sale!
Sale!












Reviews
There are no reviews yet.